 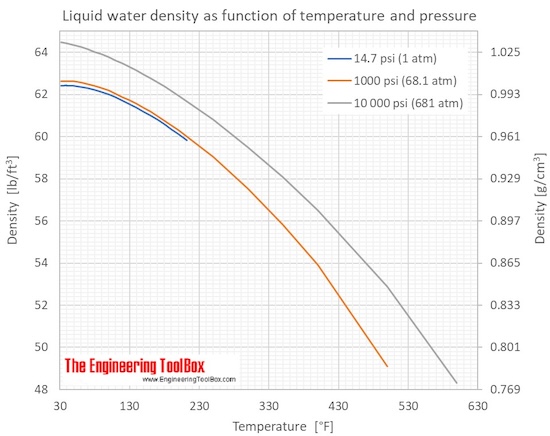 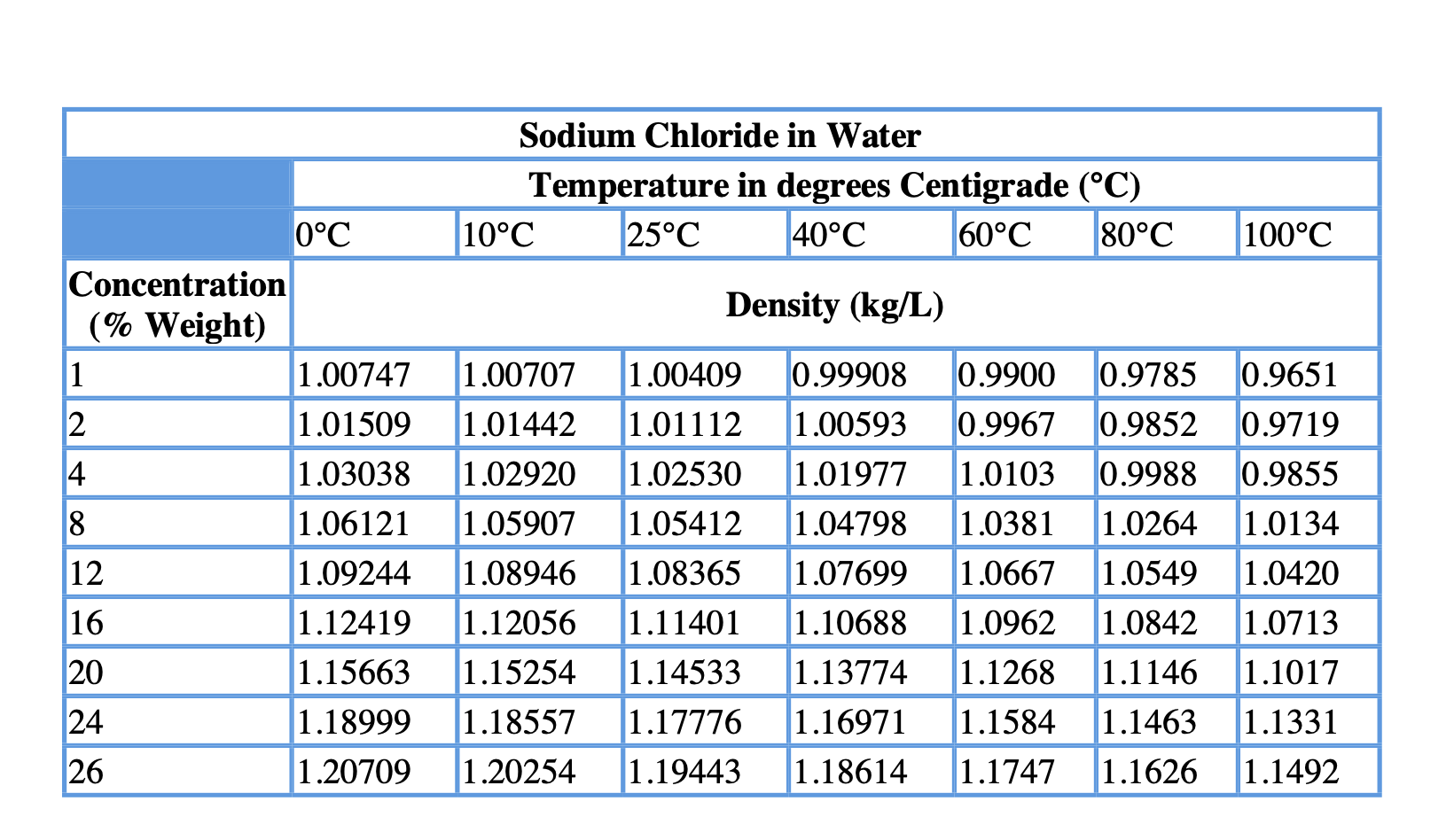 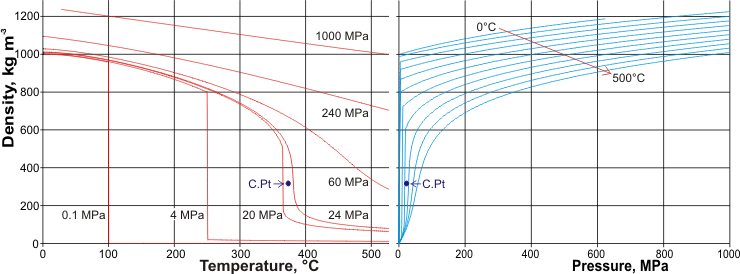
Pruß: The IAPWS Formulation 1995 for the Thermodynamic Properties of Ordinary Water Substance for General and Scientific Use. The density of water (H 2 O) changes depending on temperature and pressure - as with other substances. * - NIST Standard Reference Database 69: NIST Chemistry WebBook, Thermophysical Properties of Fluid Systems, Isobaric Properties for Water, 2008. * Temperature according to ITS-90 (International Temperature Scale from 1990). The following table lists the density D of water as a function of the temperature under isobaric conditions : This behavior of the density of water explains why ice forms at the top of a body of water. The reason why water does not possess an absolute density is because water’s density changes with temperature.Īnd there you have everything you need to know about the density of water, the factors affecting it, and how to calculate it.The density of water (H 2O) changes depending on temperature and pressure - as with other substances. The density of water increases with decreasing temperature, reaching a maximum at 4.0 ☌, and then decreases as the temperature falls below 4.0 ☌. Why water doesn’t have an absolute density? When it freezes its density decreases by about 9%. When compared to water, ice has a lower density which is the reason why it floats. What is the reason for ice floating in water?Īs we already know, water is at its maximum density at 4☌. The density of normal water is 22° C at room temperature and it is 997.77 in kg/m3. What is the density of normal water at room temperature? The reason why water is at its maximum density at 4☌ is that at 4☌ both opposing effects are in balance. Why does water have its maximum density at 4☌? Make sure to use the units of volume and mass correctly.įrequently Asked Questions About Density Of Water 1.Find out the density by dividing the mass of that liquid by volume.Calculate the mass of liquid and subtract the weight of glass from it.Handbook Of Chemistry and Physics Online. Find the weight of glassware with liquid. Click the help icon for more information on the differences between Individual User accounts and Workspace accounts.Pour liquid in it and measure the volume.As it contains two alcohol groups, it is classed as a diol.It is miscible with a broad range of solvents, including water, acetone, and chloroform. The lines are calibrated by using the hydrometer in a standard liquid with known density (usually water with a specific gravity of 1.000 at 4☌). Propylene glycol (IUPAC name: propane-1,2-diol) is a viscous, colorless liquid, which is nearly odorless but possesses a faintly sweet taste.Its chemical formula is CH 3 CH(OH)CH 2 OH. The lower it sinks, the lower is the density, the higher it floats, the higher the density. A hydrometer has a weighted bulb and a cylindrical stem with lines on it that represent how far the weighted bulb sinks in the liquid. The simplest way to find the density of a liquid is with a hydrometer. How To Calculate The Density Of A Liquid? Therefore, heavy ice sinks in normal water and floats in heavy water. When a neutron is added to an atom of hydrogen, it makes deuterium 10.6% denser than your normal water. Deuterium is an isotope of hydrogen in which there is one electron, one proton, and one neutron. Ice of heavy water sinks in water owing to its higher density because deuterium atoms replace hydrogen items in heavy water. The reason for this unusual negative thermal expansion is because of orientation-dependent, and strong, intermolecular forces, and they’re usually seen in molten silica. If you cool it down, even more, it expands and becomes less dense. to calculate the density of water, in the temperature range 5 to 40 ☌ on ITS-90, used in the gravimetric determina- tion of the volume of volumetric standards.Normally, when you cool a liquid from room temperature, it becomes denser, but with pure water, it reaches its maximum density when it is approximately at 4☌.This relation is unimodal and non-linear in nature. Water’s density is dependent on temperature.What Factors Affect The Density Of Water? So, here’s a handy table for you that tells you the density of water pounds per cubic foot or density of water in pounds along with others: Temperature Density Of Water By Temperature And WeightĪs mentioned before, the density of water can change with temperature. Plus, you can supercool water it will still remain a liquid even though it is way below its freezing point. The density of 100% pure water is actually less than 1 g/cm³. The common unit of density of water is 1 gram per cubic centimeter (1 g/cm³). The value that we usually use in mathematical calculations is 1 gram per milliliter (1 g/ml) which is also known as the fluid density of water. The density of water is defined as the weight of water (in grams) per unit volume (in milliliter) and it relies on its temperature. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
